Chemistry often involves formulas that represent complex substances and reactions. One expression that may appear in chemical discussions is x3po4, which is commonly related to compounds containing the phosphate group. In chemistry, formulas help scientists describe how atoms combine to form substances with unique properties and uses. Understanding x3po4 requires exploring the role of phosphate compounds, molecular structures, and chemical reactions. These compounds are widely used in agriculture, industry, and biological systems. By learning about x3po4 and its chemical context, readers can gain a clearer understanding of how phosphate based substances contribute to scientific research and practical applications.
The Chemical Meaning Behind X3PO4
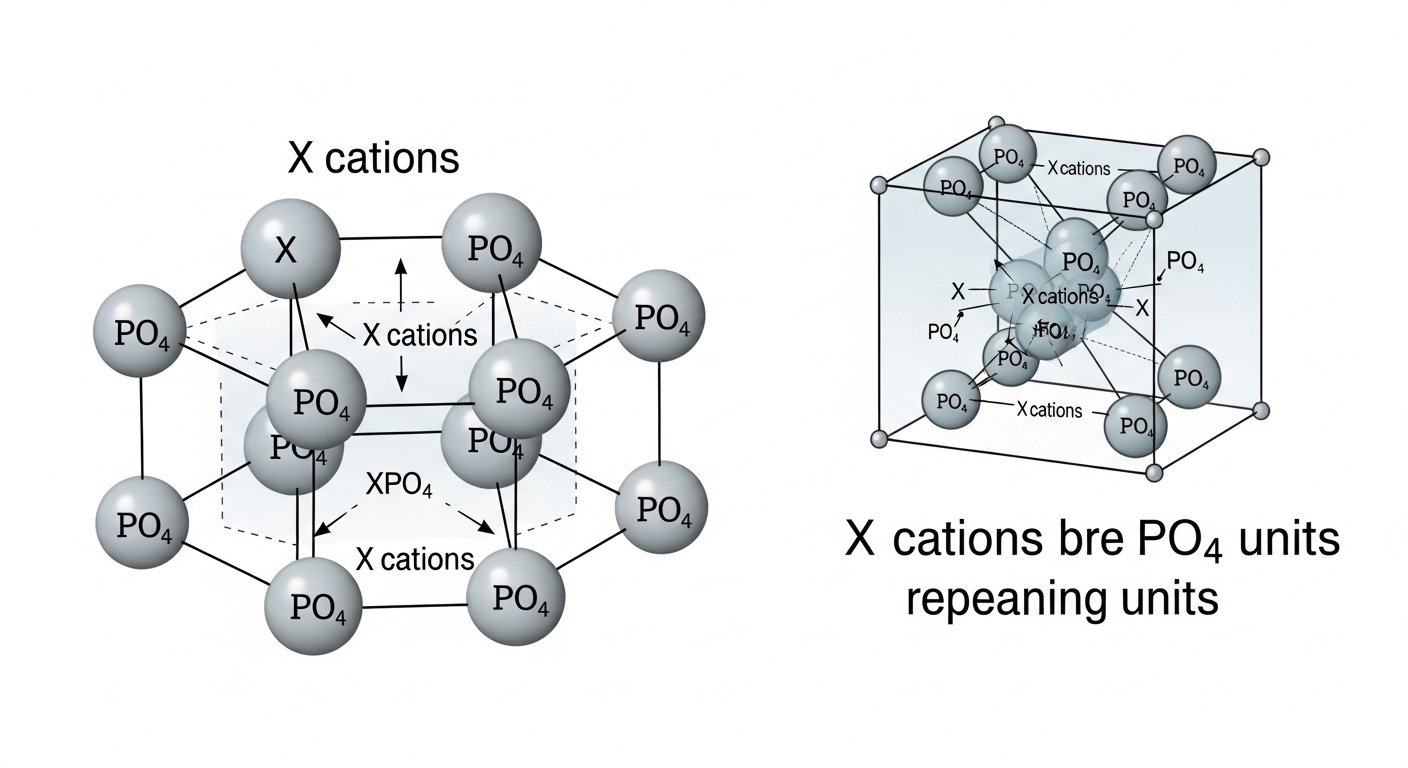
The term x3po4 represents a generalized chemical expression used to describe compounds that contain the phosphate group combined with another element or compound represented by the variable X. In chemistry, variables are sometimes used in formulas to indicate that different metals or elements can occupy a certain position in a compound. In the case of x3po4, the phosphate component PO4 plays an important role in determining the compound’s chemical properties. These phosphate based compounds are commonly found in minerals, fertilizers, and biological molecules, making them essential in both natural processes and industrial applications.
Understanding the Phosphate Group
To fully understand x3po4, it is important to explore the phosphate group, which is represented by the chemical formula PO4. This group contains one phosphorus atom bonded to four oxygen atoms, forming a stable structure with a negative charge. The phosphate group is a fundamental part of many chemical and biological compounds. It plays a vital role in energy transfer within living organisms and is also widely used in agricultural fertilizers. When the phosphate group combines with other elements, it forms compounds similar to those represented by x3po4, which can vary in structure depending on the specific element involved.
Structure and Bonding in Phosphate Compounds
The structure of compounds related to x3po4 is determined by how the phosphate group bonds with other atoms or ions. Phosphorus forms strong covalent bonds with oxygen atoms, creating a tetrahedral structure that is stable and reactive in certain conditions. When the phosphate group combines with metals or other elements represented by the variable X, ionic or covalent interactions may form. These bonding patterns influence the compound’s physical and chemical properties. Understanding the structure behind x3po4 helps chemists predict how such compounds behave in reactions, how they dissolve in water, and how they interact with other substances.
Occurrence of Phosphate Compounds in Nature
Compounds related to x3po4 occur naturally in many environments, especially in minerals and biological systems. Phosphate minerals are commonly found in rocks and soil, where they serve as important sources of phosphorus for plants. In biological systems, phosphate compounds play a key role in molecules such as DNA, RNA, and cellular energy carriers. The presence of the phosphate group in these molecules demonstrates its significance in life processes. When scientists refer to formulas like x3po4, they often study compounds that contribute to nutrient cycles and biological functions in ecosystems and living organisms.
Industrial Applications of Phosphate Compounds
Many compounds related to x3po4 have important industrial uses due to the versatile properties of phosphate groups. In agriculture, phosphate based fertilizers help plants grow by supplying essential nutrients that support root development and energy transfer. In manufacturing, phosphate compounds are used in detergents, food processing, and water treatment. Their chemical stability and ability to interact with metals make them valuable in various industrial processes. By studying structures similar to x3po4, chemists can develop new materials and improve existing products that rely on the chemical behavior of phosphate compounds.
Role in Biological Energy Systems
The phosphate group associated with x3po4 plays a central role in biological energy systems. Living cells rely on molecules that contain phosphate groups to store and transfer energy during metabolic reactions. These molecules enable cells to perform essential tasks such as growth, repair, and movement. The transfer of phosphate groups between molecules releases energy that powers many biological processes. This concept highlights the importance of phosphate chemistry in maintaining life. Understanding chemical structures related to x3po4 helps scientists study energy cycles and metabolic pathways that sustain living organisms.
Importance in Chemical Research and Education
Chemical expressions such as x3po4 are valuable in research and education because they help scientists represent generalized chemical relationships. By using variables within formulas, researchers can study entire groups of compounds instead of focusing on only one specific substance. This approach allows chemists to analyze patterns in chemical behavior and predict how similar compounds might react under different conditions. Students learning chemistry also benefit from understanding these generalized formulas because they illustrate how chemical structures can vary while still sharing common features, such as the phosphate group found in compounds related to x3po4.
Environmental Impact of Phosphate Compounds
Phosphate compounds connected to x3po4 can influence environmental systems in both positive and negative ways. On one hand, phosphorus is an essential nutrient that supports plant growth and agricultural productivity. On the other hand, excessive phosphate runoff from fertilizers can enter rivers and lakes, causing environmental problems such as algal blooms. These blooms may reduce oxygen levels in water and harm aquatic life. Understanding the chemistry of phosphate compounds helps scientists develop better strategies for managing nutrient use and protecting natural ecosystems while maintaining agricultural efficiency.
Future Developments in Phosphate Chemistry
Research related to x3po4 and phosphate chemistry continues to evolve as scientists explore new materials and applications. Advances in chemical technology allow researchers to design compounds with improved efficiency for agriculture, medicine, and environmental management. Scientists are also studying ways to recycle and conserve phosphorus resources because they are essential for global food production. As chemical research progresses, the understanding of phosphate based structures like x3po4 will contribute to innovative solutions that support sustainable development, resource conservation, and scientific discovery across multiple fields of study.
Conclusion
The concept of x3po4 highlights the importance of phosphate chemistry in both natural and industrial systems. By representing compounds that contain the phosphate group, this formula helps scientists study a wide range of chemical structures and reactions. Phosphate compounds play vital roles in agriculture, biology, and manufacturing, demonstrating their widespread significance. Understanding the structure, behavior, and applications of these compounds allows researchers to develop new technologies and manage environmental challenges more effectively. As scientific knowledge grows, the study of phosphate based formulas such as x3po4 will continue to contribute to important discoveries and practical innovations.